|
Unfortunately I cannot follow what is meant with "Entropy-Flux". Where $J_s$, $J_h$ and $J_p$ denotes the entropy flux, internal energy flux and particle flux. The differential Shannon entropy of information theory can change under a change of variables (coordinates), but the thermodynamic entropy of a physical system. Next the author presents an equation where I cannot follow in detail: I would understand this as a relation describing equilibrium states which are close together. $$T_i \delta S_i = \delta U_i - \mu_i \delta N_i$$ Entropy is a concept applied across physics, information theory, mathematics and other branches of science and engineering. On page 6 a system is split into a set of subsystems, each in local equilibrium. I just want to understand the concepts the author uses to derive some features. My question is general and not related to thermoelectricity yet. When I started with Chapter 5 "Irreversible Thermodynamics" I struggle totally with concepts of "Entropy Flow". Where n and m are the coefficients found in the balanced chemical equation of the reaction.Recently a found a paper on the thermoelectric effect: The entropy change of a reaction where the reactants and products are in their standard state can be determined using the following equation: (Source: UC Davis ChemWiki by University of California\CC-BY-SA-3.0) Standard Entropy Change of a Reaction, Δ S° Temperature of a Single Substance.” This is a generalized plot of entropy versus temperature for a single substance. These large increases occur due to sudden increased molecular mobility and larger available volumes associated with the phase changes.įigure 18.3 “Entropy vs. 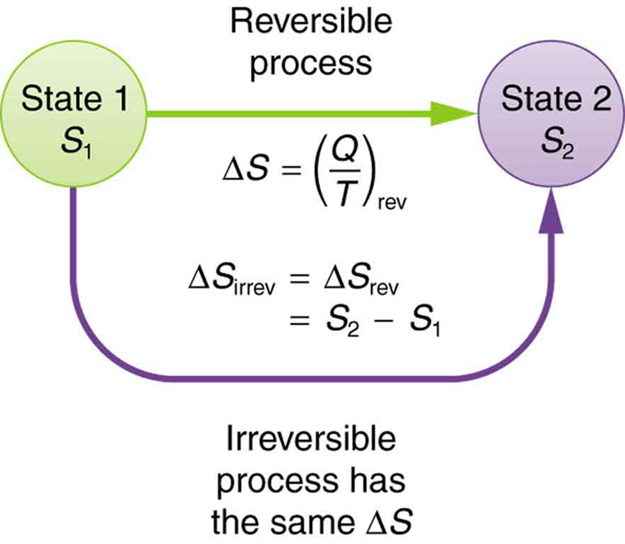
This can be seen in Figure 18.3 “Entropy vs. Temperature of a Single Substance.” Large jumps in entropy occur at the phase changes: solid to liquid and liquid to gas. As shown below, the entropy provides this measure. It has been observed in several antibody-hapten systems that maturation of immune responses leads to an increase in the association constants of induced antibodies ( Eisen and Siskind, 1964 ). , that gives an appropriate measure of the randomness of a system. The standard molar entropy of any substance increases as the temperature increases. The lock-and-key model for antibodyantigen association must be modulated by these descriptions of conformational change.Gases tend to have much larger standard molar enthalpies than liquids, and liquids tend to have larger values than solids, when comparing the same or similar substances.There are more possible arrangements of atoms in space for larger, more complex molecules, increasing the number of possible microstates. Larger, more complex molecules have higher standard molar enthalpy values than smaller or simpler molecules.Several trends emerge from standard molar entropy data: Table 18.1c Standard Molar Entropies of Selected Solids at 298 K Solid If qk is not None, then compute the relative entropy D sum(pk log(pk / qk)). If only probabilities pk are given, the Shannon entropy is calculated as H -sum(pk log(pk)). 
Table 18.1b Standard Molar Entropies of Selected Liquids at 298 K Liquid entropy (pk, qk None, base None, axis 0) source Calculate the Shannon entropy/relative entropy of given distribution(s). 
Table 18.1a Standard Molar Entropies of Selected Gases at 298 K Gas If the process takes place over a range of temperature, the quantity can be evaluated by adding bits of entropies at various temperatures. Thus, entropy has the units of energy unit per Kelvin, J K -1. These values have been tabulated, and selected substances are listed in Table 18.1a to c “Standard Molar Entropies of Selected Substances at 298 K”. Entropy is the amount of energy transferred divided by the temperature at which the process takes place. The standard molar entropy, S°, is the entropy of 1 mole of a substance in its standard state, at 1 atm of pressure. The simulation results also confirm that the proposed encryption scheme has high key security and can protect against various attacks. Assume the change is reversible and the temperature remains constant. The entropy value of the encrypted image is 7.6227, whereas the entropy value of the merge image with two input images is 3.2886, which greatly reduces the relevance of the image. The more disordered a system is, the higher (the more positive) the value of. Determine the change in entropy (in J/K) of water when 425 kJ of heat is applied to it at 50☌. Entropy can be defined as the randomness or dispersal of energy of a system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
